Advances in Power Supplies for Automated Electrochemical Mini-Plants
September 7, 2007
On-site and on-demand production of disinfectants, biocides and water purification chemicals including sodium hypochlorite and chlorine dioxide has been substantially improved via the use of advanced switch-mode power supplies that provide the power for automated electrochemical generators.
Many municipal water, food processing, and wastewater treatment plants are switching over from the use of chlorine disinfectants and biocides to safer and more environmentally friendly point-of-use and on-demand generated chemicals. The primary reasons for this change are that conventional chlorine agents require transport by tankers on accident-prone highways or railroads; there are ever increasing safety and environmental regulations regarding toxic gases and chemical spills, and these hazardous materials require bulk storage at the sites where they are used. Safer, and in many cases more effective, chemicals have been developed that can replace chlorine. For example, after many trial and error attempts to find a way to effectively control Legionnaires’ disease, it was found that chlorine dioxide (CIO2) was one of the few chemical agents that could consistently and safely disinfect Legionella bacteria (see References). Add to this the ability to manufacture these safer chemicals at the locations that use them, and only when needed, and the advantages in total become obvious.
Two popular substitute chemicals for chlorine are sodium hypochlorite (NaOCI) and chlorine dioxide (CIO2) both of which can be manufactured via mini-plants (aka, generators) that are delivered to the end users’ site as a complete package and provide the disinfectants on-demand and as needed. In many cases, these mini-plants operate automatically and can be employed in unmanned locations such as municipal water treatment sites.

These electrochemical generators use the process of electrolysis as the basis for the production of these disinfecting and biocide chemicals. Recalling our science classes, electrolysis is a common method of separating bonded elements and compounds by passing an electric current through them. It involves applying a voltage between two electrodes (anode and cathode) which are submerged in a conductive solution (electrolyte). When a voltage is applied to the electrodes, electric current flows and in turn breaks down the molecules within the solution into its components (Figure 1).
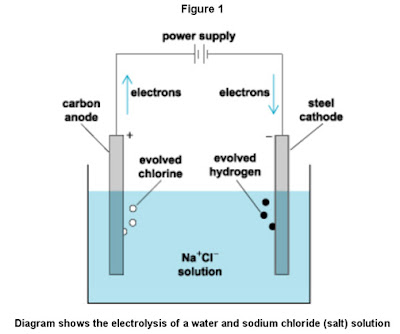
Figure 1 shows part of the process that is used to produce sodium hypochlorite (NaOCI), which is more commonly known as household bleach when sold as a solution containing 5-6% of NaOCI. However, instead of a static vessel as shown in Figure 1, modern electrochemical generators pump the electrolyte solution continuously through one or more tubes that have the electrodes mounted within them. As the electrolytic solution flows through these tubes (electrolytic cells), the electrolysis process continuously separates the molecular components. In some instances, the solution is run through the dual-electrode electrolytic cells more than once to further refine and separate the resulting chemicals. (Note: Batteries operate by a reverse process from electrolysis; they generate electricity by means of galvanic or voltaic cells that contain anode and cathode electrodes that are in contact with an electrolyte solution or gel.)
Historically, the power supplies that provide the driving force for electrochemical generators have evolved from basic transformer and diode rectifiers, to transformer and SCR (silicon-controlled-rectifier) power sources, to modern and more sophisticated power sources. The development of the switchmode power supply greatly reduced the size and substantially improved the efficiency of these power sources. In addition, switchmode power supplies have the ability to provide electronic signals for status information (volts, amps, temperature, etc.), remote control, and communications to and from a PLC (Programmable Logic Controller) or a local/remote computerized controller.
The vast majority of switchmode power supplies are designed to operate as regulated voltage power sources. These supplies regulate the output voltage very precisely regardless of the amount of current drawn from the supply, up to its design limit. For example, a 1500-watt supply can provide a 12-volts output while providing from 0 to 125 amperes of current. Once the maximum current of 125-amps is reached, the supply is designed to go into a current-limit mode (where the output voltage is automatically reduced or the supply shuts down).
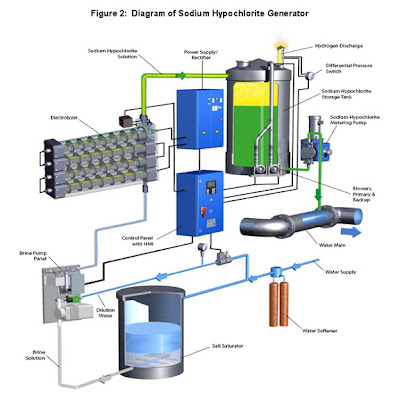
Figure 2 above shows the loop diagram of a typical sodium hypochlorite generator. The part of the system shown above that is called the Electrolyzer consists of multiple electrolytic cells (tubes containing electrodes), connected in series, through which the electrolytic solution is pumped and in turn separated into its primary chemical components (e.g., sodium hypochlorite solution and hydrogen gas) via electrolysis. The electrochemical process for manufacturing chlorine dioxide is similar to the above except it starts with a solution of sodium chlorite.
It has been found that in many electrochemical processes, including the production of disinfecting agents, that standard voltage-regulated power supplies do not always provide the ideal power profile for these processes. In fact, in many instances, the power supplies are being forced to operate at a fixed voltage and at close to their maximum current rating. If these operating conditions are maintained for long periods of time, the supply will internally heat-up and prematurely fail, thus shutting down the production of the disinfecting agents.
Why does this happen and how can it be avoided? As described above, during the electrochemical process, in order to keep up with the continuous electrolysis process with constantly flowing electrolyte solutions, the power supply must provide a high enough voltage to overcome the impedance between the two electrodes and the solution surrounding them, and, more importantly, to provide a high enough current density (amperes) to effectively separate the molecules during the short time (determined by the flow rate) that the solution comes in contact with the electrodes. By using a switchmode power supply that is designed to operate in a “constant-current” mode (instead of constant-voltage, as is the norm) the electrochemical process has been found to produce chemicals much faster, with consistent high quality, without forcing the power supply into an overload state.
There are a number of ways of providing current-mode power supplies for enhanced electrochemical applications. One method is to use Programmable Power Supplies. These supplies are designed to be manually or remotely programmed to operate in a voltage-mode and/or a current mode, at a specific voltage and current range, along with other specified parameters. As an added bonus these supplies usually include a serial digital communications port that allows it “talk” to local or remote computer controllers. Additionally, these supplies can be connected in parallel to the electrodes, or to groups of electrodes, when an electrochemical process requires more current than one supply can provide. For example, Lambda Americas’ model ZUP10-80/U programmable power supplies is adjustable from 0 to 10-volts with 0 to 80-amps (800 watts total). This type of supply is being used in its “constant-current” mode to efficiently produce disinfectant and biocide chemicals at many unmanned, non-air conditioned, municipal water treatment sites. In some applications, two or more ZUP supplies are connected in parallel to provide the necessary amount of current for the electrochemical process.

Another method of providing a “constant-current” mode power supply is to modify the design of a voltage regulated supply. This can be done by adding circuits that monitor the supply’s output current to prevent an overload, yet maintain a “constant-current” profile from the supply. For example, Lambda Americas has produced modified versions of its HWS-CC 1500-watt supply to do exactly this. In electrochemical applications that produce disinfectant and biocide chemicals, a number of these “current-mode” supplies are connected to different sets of electrodes, and/or, in parallel, to support different output current requirements for various models of electrochemical generators. Obviously, generators that produce higher output rates of chemicals require higher current levels.

This paper has focused on the techniques and benefits related to advanced switchmode power supplies for mini-electrochemical generators (self-contained plants) that produce disinfecting and biocide chemicals on-site. It should be noted that electrolysis processes are used extensively in many other chemical and industrial areas, some of which are listed below.
Many of these electrochemical processes require power levels that far exceed the range of the switchmode power supplies described above. These high power rectifier systems (ranging from 300 to 30,000 kW) are very specialized, large, heavy, and are usually comprised of huge transformers, rectifiers, thyristors, SCRs, capacitors, regulating controllers, and water cooling systems. Some of these high power sources are as large as a typical bathroom, kitchen, and larger. There is no doubt that as technologies advance, these huge power sources will see reductions in size and improvements in efficiencies.
In summary, the application of switchmode power supplies operating in a “constant-current” mode has been shown to provide significant improvements in electrochemical self contained mini-plant generators that are used to produce disinfecting and biocide chemicals. These benefits include: